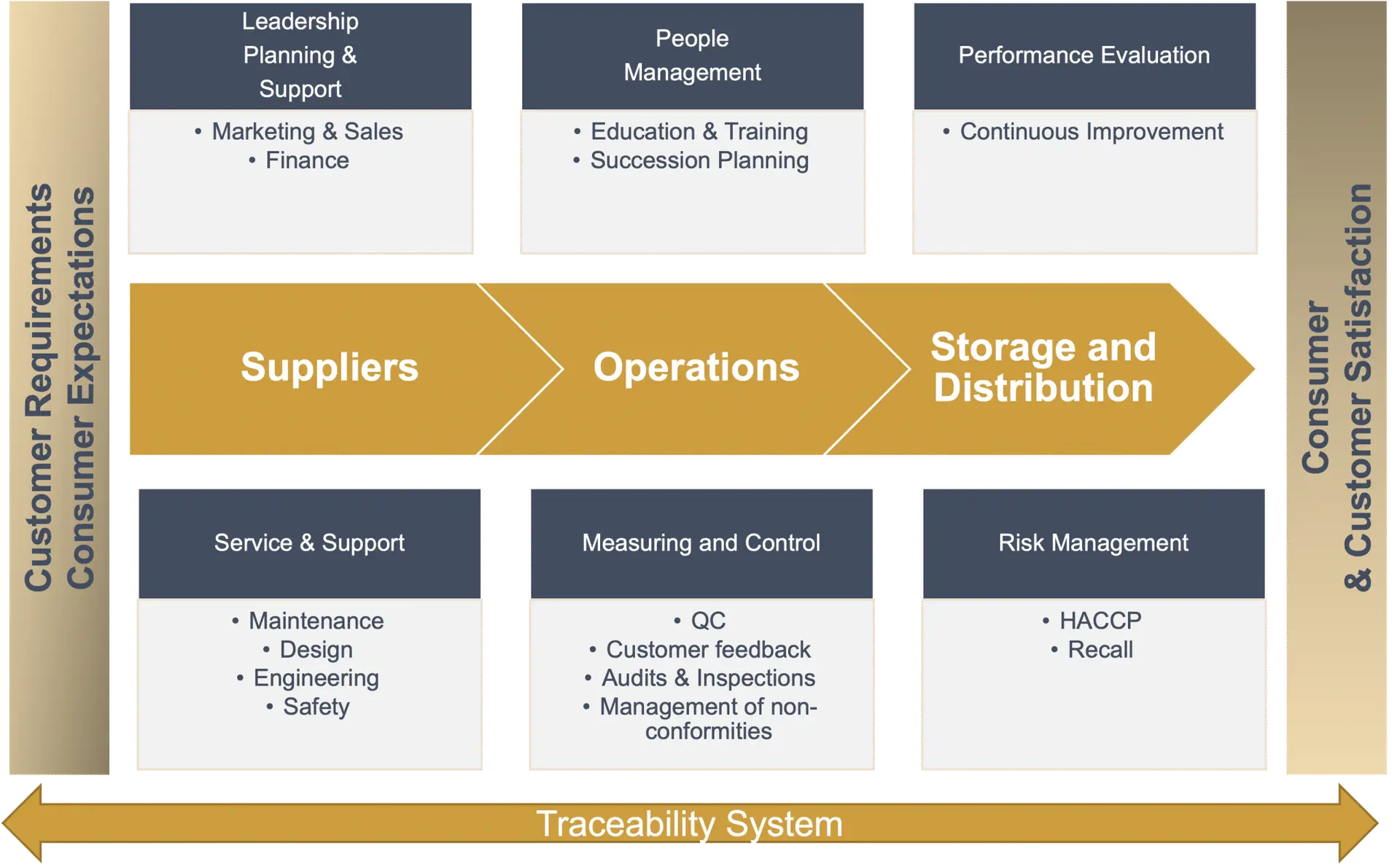
ERP implementations are a challenging but valuable investment for Food Processors and Distributors. We look at a fully successful ERP implementation as one in which:
- Master data has been fully mapped and converted
- Operational transactions such as sales orders, purchase orders, production orders, inventory moves, etc are fully enabled in the software
- Financial records including a perpetual inventory valuation, costs, etc. are accurately feeding an income statement, balance sheet, cash flow, etc.
- Business analytics with key performance metrics are in place and under regular review to drive business improvement
- Food safety controls, reporting, recall management, and compliance support are fully in place
The initial implementation of ERP within a Food Company typically achieves items 1-3 but quite often falls short with the business analytics and food safety components. We regularly encounter food companies with ERP solutions such as Aptean’s JustFood ERP, Microsoft’s Business Central, or Columbus Food and Beverage that have a working system yet their entire Food Safety and Quality (FS-Q) Management system is operating outside of ERP. As the FDA continues to move towards requiring systematized lot tracking with supply chain integration, rapid trace and recall, and immediate compliance support a non-integrated FS-Q is no longer feasible.
Here some advice and basic steps that may help you to plan the transition of FS-Q to your ERP.
An obvious prerequisite is to ensure that your current FS-Q program is effective and in compliance. If there are concerns, Adroit’s Food Safety team members can review the program and provide advice to improve the effectiveness and compliance. Once your comfortable that your system is fully in compliance with the FSMA Final Rule for Preventive Controls (3) and/or with the principles of the Hazard Analysis and Critical Control Point (HACCP) system adopted by the Codex Alimentarius Commission then you should complete an assessment of your food safety team to ensure that you have qualified food safety professionals (2).
Some key symptoms that your stand along FS-Q program is non scalable include:
- Too much data with an inability to ascertain trends or to gain useful insights
- Effective data collection but a detected increase in non-conformities
- Corrective actions are being defined and implemented upon, but it is difficult to verify the effectiveness of the corrective actions
- The traceability approach works but the reality is that executing the trace to ensure your control points and critical control points (CCPs) are under control takes too long
If your management team is increasingly uncomfortable that they really don’t know where their vulnerabilities are due to lack of information that is a symptom that your operation is growing. The amount of data being captured exceeds what you can do with manual paper-based records which are typed on spreadsheets; it is time to eliminate redundancies, automate data capture, ensuring it is traceable and that it is interconnected at different stages of the supply chain.
A good starting point is to identify common business processes like the traceability program, flow diagram, prerequisites and mandatory or non-negotiable requirements described on your HACCP or Food Safety Plan.
- Step One: Identify the site processes and flows that are site-specific and will have impact in the overall business. You can find this information in your HACCP flow diagram and/or organizational chart.
- Step Two: Identify mandatory or non-negotiable requirements, its controls and establish the criteria when the results are out of the specification. This includes HACCP hazard analysis, list of prerequisites, product description, specifications, master plan, CCPs, monitoring and verification activities.
- Purchasing
- Supplier management
- Criteria for the supplier approval and ongoing approval process
- Receiving
- Criteria based on the risk assessment of suppliers and hazard analysis to raw materials and ingredients
- Pre-receipt inventory and entry compliance criteria
- Determine key identifiers to ensure materials, processes and controls are traceable
- Batch/lot assignation is based on expiration and receiving dates, claims, allergen content, etc.
- Process Control
- Critical to food safety and quality control points
- Identification of work in progress, rework, restricted materials
- Sampling plans, process specification, which are the control limits and what decisions are made when the process and/or product is out of the spec.
- Storage and Distribution
- Lot tracking
- Identification of restricted and released materials and products, how to treat positive releasing, and how to ensure the product is identified when is required to send it out to third party storage units
- Customer Communication
- Capturing input from customers and consumers and how to respond to them
- Quality audits and checks
- From reception to in-process to finish product to shipment
- Controlling release and delivery of goods based on FS-Q audits
- Control of nonconformities
- Quality hold management (reactive approach)
- Corrective and Preventive Action
- Assigning the right SMEs for its management, root cause analysis, tracking and trending to avoid re-occurrences (proactive approach)
- Document and Records Control Management
- Identifying what data needs to be collected
- Assigning roles for monitoring and verifying activities
- Purchasing
To act on data, you first must collect it properly, ensuring traceable data will be transformed into proper insights. Better data means better performance. (5)
- Step Three: Selecting the adequate Food Safety Enterprise Resource Planning (ERP) could be a full-time project. You want to have a friendly and flexible system to ensure its sustainability over time.
- It is important that the right Subject Matter Experts (SMEs) are selected to provide business knowledge, input, and expertise to ensure legal and regulatory business requirements are captured and understood.
- Include food safety and quality (FS-Q) professionals in the earlier stages, before selecting the ERP, to guarantee every function considers key FS-Q requirements. These are always traceable.
Adroit can help you to make the best decisions, ensuring your Food Safety Plan is ERP ready, and ensuring the ERP will be adjusted to your system and business. We are experts in Aptean Food and Beverage ERP/JustFood and Microsoft Business Central.
References:
- HACCP – HAZARD ANALYSIS AND CRITICAL CONTROL POINT (HACCP) SYSTEM AND GUIDELINES FOR ITS APPLICATION – Annex to CAC/RCP 1-1969, Rev. 3 (1997)
- PCQI – Preventive Controls Qualified Individual – The Food Safety Preventive Controls Alliance (FSPCA)
- US FDA – FSMA Final Rule for Preventive Controls for Human Food
- ADROIT NA– APTEAN ERP – White Paper: Why Your Food Company Needs a Food Specific ERP Solution
- APTEAN – Food and Beverage ERP justfood Edition
Disclaimer: This information is to be used as guideline only. It is the responsibility of the readers to maintain strict compliance with all local, state and federal laws, rules, regulations and third-party standards.